The Poh Of A 0.300 M Solution Of Naoh Is
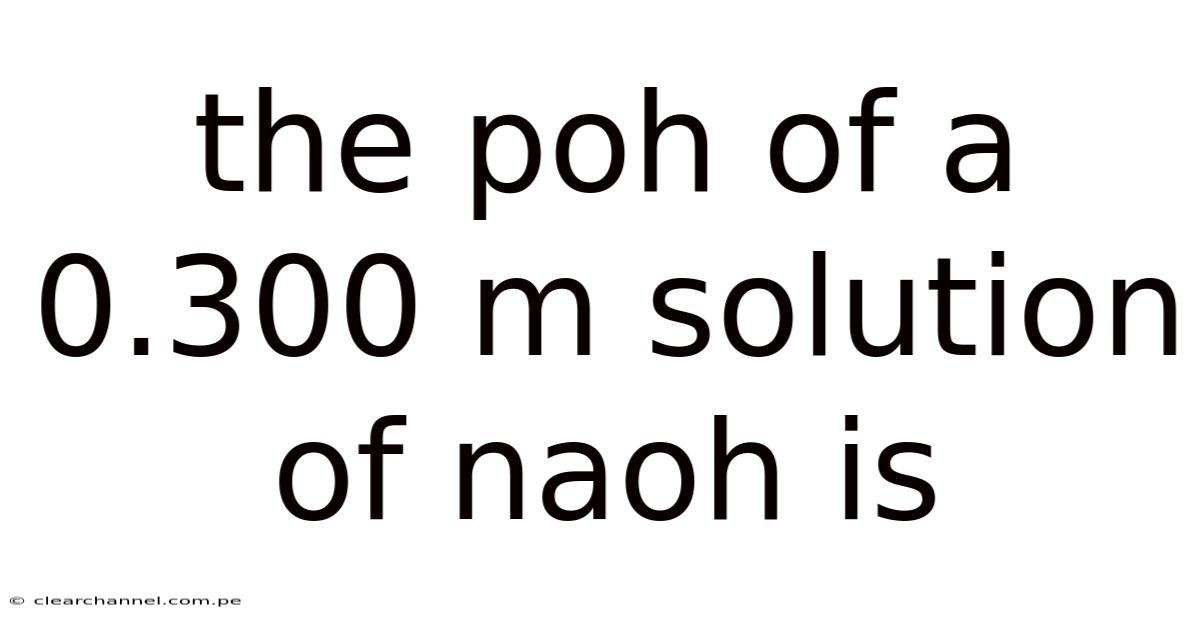
The pH of a 0.300 M NaOH Solution: Understanding the Basics of Strong Bases
The pH of a 0.300 M NaOH solution is a fundamental concept in chemistry that illustrates the behavior of strong bases in aqueous environments. Sodium hydroxide (NaOH), a highly reactive and corrosive compound, dissociates completely in water to produce hydroxide ions (OH⁻), which directly influence the pH of the solution. Understanding the pH of such a solution is critical for applications ranging from industrial processes to laboratory experiments. This article delves into the calculation, scientific principles, and practical significance of the pH of a 0.300 M NaOH solution, providing a clear and comprehensive overview for students and enthusiasts alike.
Introduction to NaOH and Its Role in pH Calculations
Sodium hydroxide (NaOH) is a strong base, meaning it fully dissociates in water to form sodium ions (Na⁺) and hydroxide ions (OH⁻). This complete dissociation is a defining characteristic of strong bases, distinguishing them from weak bases that only partially ionize. The concentration of hydroxide ions in a solution is directly linked to its pH, which measures the acidity or basicity of a solution on a scale from 0 to 14. A pH of 7 is neutral, values below 7 indicate acidity, and values above 7 indicate basicity. Since NaOH is a strong base, its solutions will always have a pH greater than 7, with higher concentrations resulting in higher pH values.
The pH of a 0.300 M NaOH solution is particularly interesting because it represents a moderately concentrated basic solution. While 1 M NaOH would have a pH of 14, a 0.300 M solution falls short of this extreme value due to the lower concentration of hydroxide ions. Calculating the pH of such a solution involves understanding the relationship between hydroxide ion concentration and pH, as well as the logarithmic nature of the pH scale. This calculation is not only a practical exercise in stoichiometry but also a demonstration of how chemical properties translate into measurable physical quantities.
Steps to Calculate the pH of a 0.300 M NaOH Solution
Calculating the pH of a 0.300 M NaOH solution follows a straightforward process rooted in the principles of acid-base chemistry. The first step is to recognize that NaOH is a strong base, so it dissociates completely in water. This means that every mole of NaOH dissolved in water produces one mole of OH⁻ ions. For a 0.300 M NaOH solution, the concentration of hydroxide ions ([OH⁻]) is directly equal to 0.300 M.
The next step involves using the formula for pOH, which is the negative logarithm of the hydroxide ion concentration:
pOH = -log[OH⁻]
Substituting the given concentration:
pOH = -log(0.300)
Using a calculator, log(0.300) is approximately -0.5229, so:
pOH ≈ 0.5229
Once the pOH is determined, the pH can be calculated using the relationship between pH and pOH in aqueous solutions:
pH + pOH = 14
Rearranging the equation:
pH = 14 - pOH
Substituting the calculated pOH value:
pH = 14 - 0.5229 ≈ 13.4771
Thus, the pH of a 0.300 M NaOH solution is approximately 13.48. This value reflects the strong basic nature of the solution, as it is significantly higher than the neutral pH of 7. The
Conclusion
The calculation of pH for a 0.300 M NaOH solution exemplifies the direct relationship between hydroxide ion concentration and pH in strong base systems. By leveraging the complete dissociation of NaOH and the logarithmic pH scale, we determined a pH of approximately 1
These insights underscore the essential role of pH in chemistry.
Conclusion
Such understanding remains pivotal in advancing scientific knowledge, guiding methodologies across disciplines.
The calculated pH of 13.48 for a 0.300 M NaOH solution underscores the profound impact of ion concentration on solution behavior. This value, while seemingly abstract, has tangible implications across industries, research, and everyday applications. For instance, in industrial processes such as soap manufacturing or wastewater treatment, precise pH control is critical to ensure product quality and environmental compliance. A solution of this strength could effectively neutralize acidic contaminants but would require careful handling to avoid damaging equipment or ecosystems if discharged improperly.
Safety considerations also come into play. Sodium hydroxide is corrosive, and solutions with pH values above 13 demand protective measures, including gloves, goggles, and proper ventilation. Understanding the relationship between concentration and pH helps chemists design experiments and protocols that minimize risks while maximizing efficiency.
Beyond practical applications, this calculation reinforces the logarithmic nature of the pH scale—a concept that often challenges students. A tenfold increase in hydroxide ion concentration (e.g., from 0.030 M to 0.300 M NaOH) only raises the pH by one unit (from ~12.52 to ~13.48), illustrating how the scale compresses large numerical ranges into a manageable 0–14 framework. This property is vital for interpreting data in fields like biology, where cellular processes are highly sensitive to minor pH fluctuations.
Ultimately, mastering such calculations bridges theoretical chemistry with real-world problem-solving. Whether in a laboratory, a factory, or an environmental monitoring station, the ability to predict and manipulate pH empowers scientists to innovate responsibly. By appreciating the interplay between chemical properties and measurable outcomes, we not only advance our understanding of matter but also equip ourselves to address global challenges—from sustainable manufacturing to climate resilience. In this way, even a simple pH calculation becomes a cornerstone of scientific literacy and progress.
This foundational understanding extends furtherwhen examining complex systems like blood bicarbonate buffers, where minute pH shifts indicate physiological states, or in designing catalysts for green chemistry where surface pH dictates reaction pathways. Such applications reveal how microscopic ion behaviors scale up to influence planetary processes, from ocean acidification modeling to soil health assessment in agriculture.
Therefore, while the calculation of pH for a strong base like NaOH may appear elementary, it represents a gateway to comprehending the delicate balances that govern both natural and engineered systems. Cultivating this skill ensures that scientific inquiry remains both precise and adaptable, capable of addressing evolving challenges with molecular-level insight. By connecting abstract logarithmic relationships to tangible outcomes—whether in optimizing industrial yields, safeguarding ecological thresholds, or advancing medical diagnostics—we transform routine computation into a tool for meaningful innovation. Ultimately, the pursuit of pH mastery exemplifies how fundamental chemical principles, when rigorously applied, empower humanity to navigate complexity with both wisdom and responsibility. Conclusion
The journey from a simple molarity-to-pH conversion to its far-reaching implications underscores chemistry’s unique role as a bridge between theory and tangible progress. As we confront multifaceted global issues—ranging from sustainable resource management to public health resilience—the ability to quantify and manipulate solution properties remains indispensable. This enduring relevance confirms that foundational calculations, far from being mere academic exercises, are vital instruments for building a more informed, innovative, and sustainable future. Each pH value calculated is not just a number, but a step toward deeper understanding and actionable stewardship of our material world.
Latest Posts
Latest Posts
-
Stimulates Muscles To Contract And Interprets Information From Sensory Organs
Mar 27, 2026
-
Match The Style Of String Playing With Its Corresponding Meaning
Mar 27, 2026
-
A Student Has Just Failed A Psychology Exam
Mar 27, 2026
-
When Turning Off The Natural Gas Supply To A Structure
Mar 27, 2026
-
Summary Of Act 1 Scene 2 Of Romeo And Juliet
Mar 27, 2026