In Glycolysis There Is A Net Gain Of _____ Atp.
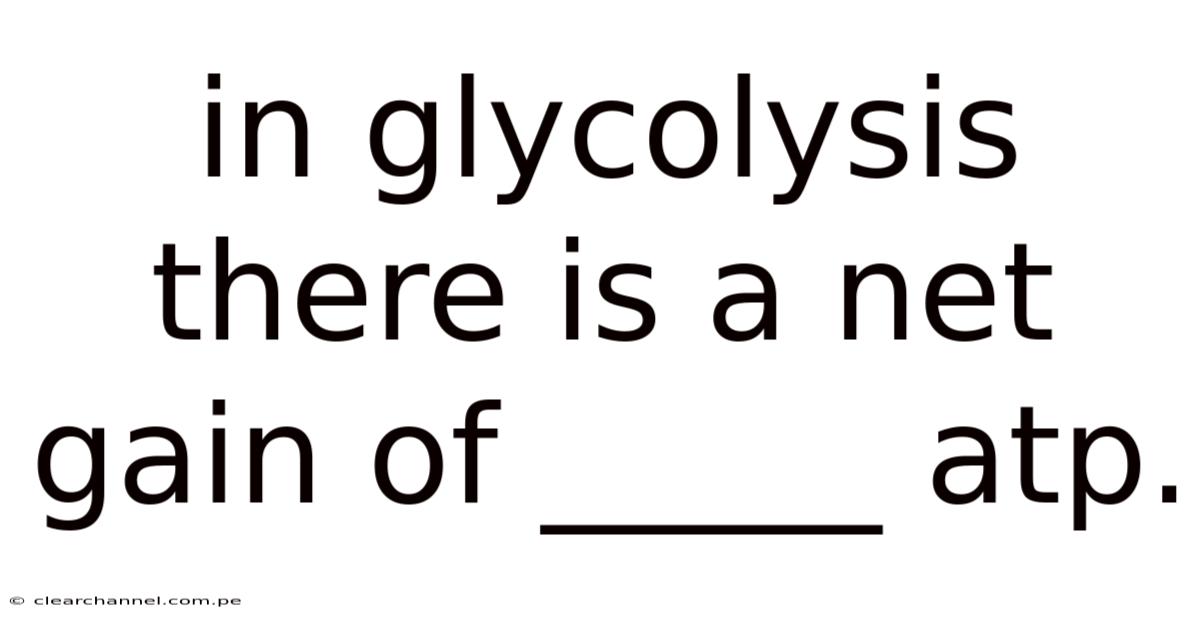
Inglycolysis there is a net gain of 2 ATP per molecule of glucose, a fundamental concept that underpins cellular energy metabolism and appears repeatedly in biochemistry curricula. Understanding why the pathway yields exactly two ATP molecules—despite consuming and producing several phosphorylated intermediates—helps students grasp how cells balance energy investment and payoff during the breakdown of sugar. This article walks through the glycolytic sequence, highlights the enzymes and substrates involved, explains the ATP accounting, and discusses the physiological relevance of the net gain.
Overview of Glycolysis
Glycolysis, derived from the Greek words glykys (sweet) and lysis (splitting), is the cytosolic pathway that converts one six‑carbon glucose molecule into two three‑carbon pyruvate molecules. The process occurs in virtually all living cells and does not require oxygen, making it a central hub for both aerobic and anaerobic metabolism. Although ten enzymatic steps comprise glycolysis, they can be conceptually divided into two phases: an energy‑investment phase that consumes ATP, and an energy‑payoff phase that generates ATP and NADH.
Steps of Glycolysis
Below is a concise, numbered list of the ten reactions, grouped by phase. Each step notes the enzyme, substrate, product, and any ATP or NADH involvement.
Energy‑Investment Phase (Steps 1‑5)
- Hexokinase (or glucokinase) phosphorylates glucose at C‑6 using ATP → glucose‑6‑phosphate + ADP.
- Phosphoglucose isomerase converts glucose‑6‑phosphate to fructose‑6‑phosphate (isomerization). 3. Phosphofructokinase‑1 (PFK‑1) adds a second phosphate from ATP to fructose‑6‑phosphate at C‑1 → fructose‑1,6‑bisphosphate + ADP.
- Aldolase cleaves fructose‑1,6‑bisphosphate into two three‑carbon sugars: dihydroxyacetone phosphate (DHAP) and glyceraldehyde‑3‑phosphate (G3P).
- Triose phosphate isomerase rapidly equilibrates DHAP with G3P, yielding two molecules of G3P for the downstream payoff phase.
Investment cost: 2 ATP molecules are consumed (steps 1 and 3).
Energy‑Payoff Phase (Steps 6‑10)
- Glyceraldehyde‑3‑phosphate dehydrogenase oxidizes each G3P, reducing NAD⁺ to NADH and attaching a phosphate → 1,3‑bisphosphoglycerate (1,3‑BPG).
- Phosphoglycerate kinase transfers the high‑energy phosphate from 1,3‑BPG to ADP, producing ATP and 3‑phosphoglycerate (3‑PG).
- Phosphoglycerate mutase relocates the phosphate from C‑3 to C‑2 → 2‑phosphoglycerate (2‑PG).
- Enolase removes a water molecule from 2‑PG, forming phosphoenolpyruvate (PEP).
- Pyruvate kinase transfers the phosphate from PEP to ADP, yielding another ATP and pyruvate.
Payoff yield: For each G3P, steps 7 and 10 generate one ATP each. Since two G3P molecules arise from one glucose, the payoff phase produces 4 ATP and 2 NADH.
Net ATP Calculation
Summing the investment and payoff phases:
- ATP consumed: 2 (hexokinase + PFK‑1)
- ATP produced: 4 (two from phosphoglycerate kinase, two from pyruvate kinase)
[ \text{Net ATP} = 4 \text{ produced} - 2 \text{ consumed} = \mathbf{2;ATP} ]
Thus, the statement “in glycolysis there is a net gain of 2 ATP” is quantitatively accurate per glucose molecule.
Role of NADH
Although the question focuses on ATP, the NADH generated in step 6 is energetically valuable. Under aerobic conditions, each cytosolic NADH can yield approximately 1.5–2.5 ATP via the mitochondrial electron transport chain (depending on the shuttle system). Consequently, the total energetic output of glycolysis can be higher when oxidative phosphorylation is functional, but the substrate‑level phosphorylation component remains fixed at 2 ATP.
Regulation of Glycolytic ATP Yield
The net ATP gain is not merely a static number; it is modulated by key regulatory enzymes that respond to cellular energy status:
- Phosphofructokinase‑1 (PFK‑1) is allosterically inhibited by high ATP and citrate, and activated by AMP and fructose‑2,6‑bisphosphate. When ATP is abundant, PFK‑1 activity drops, reducing glycolytic flux and conserving glucose.
- Pyruvate kinase is inhibited by ATP and alanine, and activated by fructose‑1,6‑bisphosphate (feed‑forward stimulation).
- Hexokinase is feedback‑inhibited by its product glucose‑6‑phosphate, preventing unnecessary phosphorylation when downstream intermediates accumulate.
These controls ensure that the net gain of 2 ATP aligns with the cell’s immediate energetic demands.
Physiological Significance
- Rapid ATP Supply – Glycolysis provides ATP within milliseconds, crucial for tissues with high, fluctuating energy needs such as erythrocytes (which lack mitochondria) and exercising skeletal muscle.
- Anabolic Precursors – Intermediates like glucose‑6‑phosphate, fructose‑6‑phosphate, and 3‑phosphoglycerate feed into pathways for nucleotide synthesis, amino acid biosynthesis, and glycerol‑3‑phosphate formation for lipid metabolism. 3. Redox Balance – The NADH produced must be reoxidized to sustain glycolysis. In anaerobic conditions, lactate dehydrogenase converts pyruvate to lactate, regenerating NAD⁺. In aerobic cells, NADH feeds into oxidative phosphorylation.
- Disease Relevance – Dysregulation of glycolytic enzymes is implicated in cancer (the Warburg effect), diabetes, and rare enzymatic deficiencies (e.g., pyruvate kinase deficiency causing hemolytic anemia). Understanding the ATP yield helps explain why cancer cells upregulate glycolysis despite its low ATP efficiency—they prioritize biosynthetic precursors over maximal ATP production.
Frequently Asked Questions
Q1: Why does glycolysis consume ATP before producing any?
A: The initial phosphorylation steps (hexokinase and PFK‑1) destabilize glucose, making it prone to cleavage and allowing subsequent steps to release enough free energy to drive ATP synthesis later. This “investment” creates high‑energy intermediates that can donate phosphate to ADP with a large negative ΔG.
Q2: Can the net ATP gain ever differ from 2?
A: In standard glycolysis of one glucose molecule, the substrate‑level phosphorylation yield is invariant at 2 ATP. Variations arise only when alternative pathways (e.g., the pentose phosphate pathway) shunt intermediates away, or when cells use glycogen as a glucose source (which bypasses the hexokinase step, slightly altering the net cost).
**Q3: How does the presence
Q3: How does the presence of oxygen influence the net ATP yield from glycolysis?
A: Oxygen itself does not directly alter the substrate-level phosphorylation yield of 2 ATP per glucose in glycolysis. However, oxygen determines the fate of pyruvate and NADH. Under aerobic conditions, pyruvate enters the mitochondria for oxidative phosphorylation, and NADH is shuttled into the electron transport chain, yielding an additional ~2.5–3 ATP per NADH (or ~5 ATP total from glycolytic NADH). Under anaerobic conditions, NADH is used to reduce pyruvate to lactate (in muscle) or ethanol (in yeast), regenerating NAD⁺ but forfeiting the potential oxidative phosphorylation yield. Thus, the total ATP derived from one glucose molecule can range from 2 (anaerobic) to approximately 7–8 (aerobic, including mitochondrial ATP from glycolytic NADH), but the glycolytic pathway itself consistently produces a net of 2 ATP.
Conclusion
Glycolysis stands as a central, highly regulated metabolic pathway that provides a rapid, albeit modest, net gain of 2 ATP per glucose molecule through substrate-level phosphorylation. Its elegance lies in its tight allosteric control, which matches glycolytic flux to cellular energy status, and its integration with broader metabolic networks. The pathway serves not only as an immediate energy source for specialized cells like erythrocytes but also as a critical supplier of carbon skeletons for biosynthesis and a regulator of redox balance via NAD⁺ regeneration. The consistent 2 ATP yield, despite variations in oxygen availability or alternative metabolic shunts, underscores glycolysis’s role as a foundational energy-producing module. Its dysregulation is a hallmark of diseases such as cancer, where the Warburg effect highlights a strategic shift toward biosynthetic precursor production over efficient ATP yield. Ultimately, glycolysis exemplifies a beautifully balanced system—conservative in its core ATP accounting yet exquisitely adaptable to the diverse and dynamic demands of eukaryotic life.
Latest Posts
Latest Posts
-
Cuticle Softeners Are Not Designed For The
Mar 19, 2026
-
The Method That Completely Destroys Microorganisms Is
Mar 19, 2026
-
The Following Descriptions Are All Benefits Of A Product Except
Mar 19, 2026
-
When Caring For A Patient With An Open Facial Injury
Mar 19, 2026
-
Which Are Appropriate Interventions For An Apneic Child
Mar 19, 2026