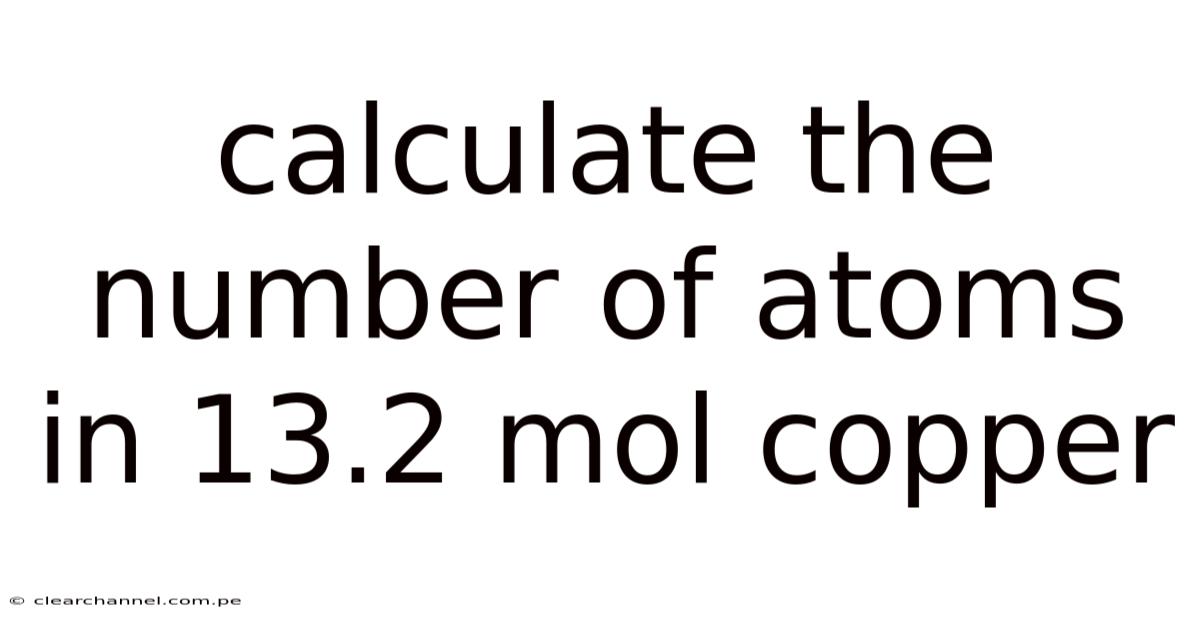Calculate the Number of Atoms in 13.2 Mol Copper: A Step-by-Step Guide
When dealing with chemical calculations, understanding how to convert between moles and atoms is fundamental. This process is essential for students, researchers, and anyone working with chemistry or related fields. The concept of moles and Avogadro’s number forms the backbone of such calculations, allowing scientists to quantify substances at the atomic level. In this article, we will explore how to calculate the number of atoms in 13.2 mol of copper, breaking down the steps, the scientific principles involved, and addressing common questions. Whether you’re a student or a curious learner, this guide will provide a clear and practical approach to mastering this calculation.
Understanding the Basics: Moles and Avogadro’s Number
Before diving into the specific calculation, it’s crucial to grasp the foundational concepts. A mole is a unit used in chemistry to measure the amount of a substance. One mole contains exactly 6.022 x 10²³ particles, whether they are atoms, molecules, or ions. This number is known as Avogadro’s number, named after the Italian scientist Amedeo Avogadro. The significance of Avogadro’s number lies in its ability to bridge the gap between the microscopic world of atoms and the macroscopic world we can measure.
For example, if you have 1 mole of copper atoms, you have 6.022 x 10²³ copper atoms. This relationship is universal, meaning it applies to any element or compound. However, the mass of 1 mole of a substance varies depending on its atomic mass. Copper, for instance, has an atomic mass of approximately 63.55 atomic mass units (amu), which means 1 mole of copper weighs 63.55 grams. While the mass differs, the number of atoms in 1 mole remains constant at Avogadro’s number.
Step-by-Step Calculation: From Moles to Atoms
Calculating the number of atoms in 13.2 mol of copper involves a straightforward multiplication using Avogadro’s number. Here’s how to do it:
- Identify the Given Value: The problem states 13.2 mol of copper. This is the amount of copper we need to convert into atoms.
- Apply Avogadro’s Number: Multiply the number of moles by Avogadro’s number (6.022 x 10²³ atoms/mol).
- Perform the Calculation:
$ 13.2 , \text{mol} \times 6.022 \times 10^{23} , \text{atoms/mol} = 7.94904 \times 10^{24} , \text{atoms} $ - Round the Result: Depending on the required precision, the result can be rounded. For most practical purposes, rounding to three significant figures is sufficient, giving $7.95 \times 10^{24}$ atoms.
This calculation demonstrates how a relatively small number of moles (13.2) can translate into an astronomically large number of atoms. The key takeaway is that Avogadro’s number allows us to scale up from moles to individual atoms efficiently.
Scientific Explanation: Why This Works
The calculation relies on the definition of a mole and the consistency of Avogadro’s number. A mole is defined as the amount of substance that contains as many elementary entities (atoms, molecules, etc.) as there are atoms in 12 grams of carbon-12. This definition ensures that Avogadro’s number is a fixed value, making it a reliable constant in chemical calculations.
In the case of copper, the atomic mass of 63.55 amu means that 1 mole of copper atoms weighs 63.55 grams. However, the number of atoms in that 1 mole is always 6.022 x 10²³, regardless of the element. This universality is what makes Avogadro’s number so powerful. When you multiply the number of moles by Avogadro’s number, you are essentially counting the total number of atoms in the given sample.
It’s also worth noting that this method applies to any element or compound. For instance, if you had 5.0 mol of oxygen molecules (O₂), you would still multiply 5.0 by Avogadro’s number to find the number of molecules. However, if you wanted the number of oxygen atoms, you would need to account for the fact that each O₂ molecule contains two oxygen atoms, requiring an additional multiplication by 2. In the case of copper, since it is an element and not a molecule, no such adjustment is necessary.
**Common
Such foundational knowledge remains integral to scientific advancement, bridging abstract principles with tangible outcomes.
Conclusion: These principles continue to serve as pillars guiding exploration and innovation across disciplines, ensuring continuous progress in both theoretical and applied realms.
Building onthis foundation, the ability to translate between macroscopic samples and microscopic counts underpins every quantitative discipline in chemistry, physics, and materials science. In industrial settings, engineers use the same conversion to design catalysts, optimize reaction yields, and ensure that quality‑control specifications are met with confidence. In the laboratory, researchers routinely employ gravimetric and titimetric analyses that begin with a precise mole calculation, then scale up to the number of particles involved, enabling reproducible results that can be compared across laboratories worldwide.
The same arithmetic also guides the development of emerging technologies such as nanotechnology and quantum materials, where the properties of a substance can change dramatically when confined to a few hundred or thousand atoms. By mastering the mole‑to‑particle relationship, scientists can predict how size, surface area, and electronic structure will evolve as a material is engineered at the atomic level, opening pathways to ultra‑lightweight composites, high‑efficiency batteries, and next‑generation electronic devices.
Educators, too, leverage this conversion as a teaching cornerstone, using it to illustrate the bridge between the tangible world we can weigh and the invisible realm of atoms that governs chemical behavior. Interactive simulations and hands‑on experiments reinforce the concept that a seemingly modest quantity—like 13.2 mol of copper—contains an astronomical number of particles, fostering a sense of wonder that fuels curiosity and scientific thinking.
In sum, the simple multiplication of moles by Avogadro’s number is more than a procedural step; it is a gateway that connects everyday measurements to the fundamental building blocks of matter. Mastery of this gateway equips scientists, engineers, and students alike with the quantitative language needed to explore, innovate, and solve the complex challenges of the modern world.
The ability to translate between macroscopic samples and microscopic counts underpins every quantitative discipline in chemistry, physics, and materials science. In industrial settings, engineers use the same conversion to design catalysts, optimize reaction yields, and ensure that quality‑control specifications are met with confidence. In the laboratory, researchers routinely employ gravimetric and titimetric analyses that begin with a precise mole calculation, then scale up to the number of particles involved, enabling reproducible results that can be compared across laboratories worldwide.
The same arithmetic also guides the development of emerging technologies such as nanotechnology and quantum materials, where the properties of a substance can change dramatically when confined to a few hundred or thousand atoms. By mastering the mole‑to‑particle relationship, scientists can predict how size, surface area, and electronic structure will evolve as a material is engineered at the atomic level, opening pathways to ultra‑lightweight composites, high‑efficiency batteries, and next‑generation electronic devices.
Educators, too, leverage this conversion as a teaching cornerstone, using it to illustrate the bridge between the tangible world we can weigh and the invisible realm of atoms that governs chemical behavior. Interactive simulations and hands‑on experiments reinforce the concept that a seemingly modest quantity—like 13.2 mol of copper—contains an astronomical number of particles, fostering a sense of wonder that fuels curiosity and scientific thinking.
In sum, the simple multiplication of moles by Avogadro’s number is more than a procedural step; it is a gateway that connects everyday measurements to the fundamental building blocks of matter. Mastery of this gateway equips scientists, engineers, and students alike with the quantitative language needed to explore, innovate, and solve the complex challenges of the modern world.